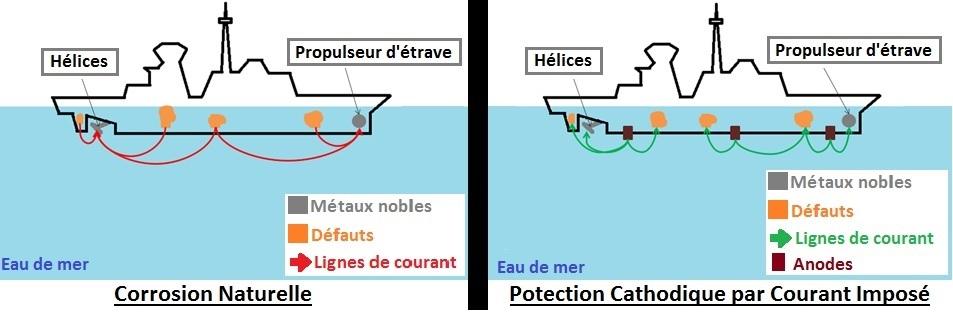
In recent decades, research for the optimization of corrosion protection systems has focused on both the reduction of energy consumption and the electromagnetic discretion of ships. Among the sources impacting the electromagnetic signature of ships, corrosion processes and corrosion protection are the most important. Representing a detection risk for naval vessels, this signature has to be reduced without comprising an optimal corrosion protection of ships. The prediction of the electromagnetic signature of ships is very often done by numerical modeling of the propagation of electromagnetic fields. The CorOns solver, based on the boundary element method, at the origin of the inter-laboratory collaboration (DGA, G2Elab and LEPMI), has been developed in this context. These researches are based on experimental studies of interfaces and their evolution according to different corrosion scenarios encountered in real conditions. The use of electrochemical impedance spectroscopy (EIS) and voltammetry, allowed to characterize the different interfaces present at the scale of the ship hull: propeller material (stainless steel and NAB) and hull (painted and unpainted carbon steel) in synthetic sea water. Studies carried out during this project show that diffusion of dissolved oxygen under cathodic polarization, is the main factor that influences current densities and thus the electrical signatures of the ships. Under open circuit conditions, EIS showed that the electrochemical behavior of painted steel and chemically passivated martensitic steel is not impacted by the variation of the physical conditions of the environment. A simple CPE//R circuit was used to modelize the protective layer formed onto the studied substrates. The normal distribution of time-constant related to the variation of the resistivity of the layers has been modeled by a power law model for the CPE [1]–[4]. The same type of circuit was used for the unpainted steel studied at different temperatures. In this case, the CPE is modeled by Brug's (surface distribution of time-constant related to a distribution of capacitance = CPE behaviors) [5]. The dynamic conditions and the state of ageing of the surface appeared as key factors for the definition of the boundary conditions in the CorOns solver. Finally, the impact of ageing was also observed on the painted samples. After 2 years of immersion, the resistance of the painting decreases by a factor 10. Whereas visually the paint didn’t show any defect, the impedance spectroscopy highlighted the observation of new corrosion process presumably due to progressive porosity of the paint deposited onto substrate. This suggests that painted steels, even without apparent defects, can no longer be considered as insulating surfaces along time in the numerical simulation approach. A non-proportional relationship between the percentage of painted free surface and the current densities was also observed under cathodic protection conditions, related to “boundary effect”. Larger scale tests on physical scale model validated the use of the polarization curves obtained in the laboratory as boundary conditions in the CorOns solver.
[1] B. Hirschorn et al., J. Electrochem. Soc., vol. 157, no. 12, p. C458, 2010,
[2] B. Hirschorn et al., J. Electrochem. Soc., vol. 157, no. 12, p. C452, 2010,
[3] B. Hirschorn et al; Electrochim. Acta, vol. 55, no. 21, pp. 6218–6227, 2010
[4] M. E. Orazem et al., J. Electrochem. Soc., vol. 160, no. 6, pp. C215–C225, 2015
[5] G. J. J. Brug et al. , J. Electroanal. Chem., vol. 176, no. 1–2, pp. 275–295, 1984
Directeurs de thèse : Virginie ROCHE, Jean-Claude LEPRETRE
| Virginie ROCHE | MAITRE DE CONFERENCE HDR | Grenoble INP | Directeur de thèse | |||
| Bernard TRIBOLLET | PROFESSEUR EMERITE | Sorbonne Université | Rapporteur | |||
| Philippe REFAIT | PROFESSEUR DES UNIVERSITES | Université de la Rochelle | Rapporteur | |||
| Olivier CHADEBEC | DIRECTEUR DE RECHERCHE | CNRS délégation Alpes | Examinateur | |||
| Olivier DEVOS | PROFESSEUR DES UNIVERSITES | Université de Bordeaux | Examinateur | |||
| Ricardo NOGUEIRA | PROFESSEUR | Khalifa University | Examinateur | |||
| Eric SAINT-AMAN | PROFESSEUR DES UNIVERSITES | Université Grenoble Alpes | Examinateur | |||
| Cédric GOEAU | INGENIEUR DOCTEUR | Techniques navales Brest | Examinateur |
Adresse de la soutenance : salle Amphi MJK


