Contact: Dr. Virginie Roche (UGA)
Amorphous materials
Since 2010, studies have been carried out on the characterization of the corrosion resistance properties of metallic glassy alloys in collaboration with Federal University of São Carlos (UFSCar), in Brazil and, in 2012, also with SIMaP laboratory, in Grenoble.
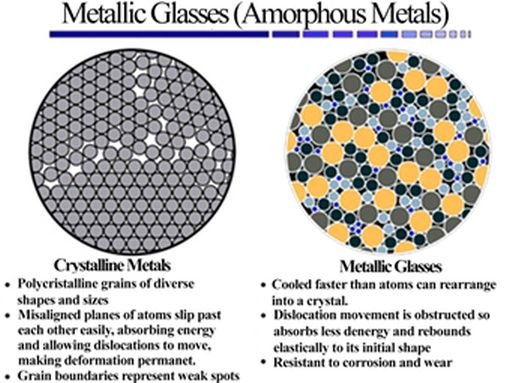
Roughly, metallic glasses are amorphous solid materials (with local ordering of the atoms, but without long-range ordering) obtained from a liquid, which does not crystallize during fast cooling (definition in figure 1).
Among the several glassy alloys that now can be produced, we firstly focused on iron-based ones for applications in coatings because of their good tribological properties and high corrosion resistance, which are due to both homogeneous composition and absence of crystalline defects that favor the formation of stable surface passivation film.
Besides, biocompatible metallic glasses based on Ti, Fe or Mg were also investigated because of their unique mechanical properties, and to study biodegradation and bioactivity. They can be used in biomedical applications (as bulk or coatings) to replace crystalline Ti-based alloys. Amorphous Ti-based alloys were studied between 2012 to 2014 in the framework of an ANR program between France and China, and in collaboration with SIMaP laboratory and two Chinese laboratories from University of Beijing. The Mg based alloys started to be studied in 2012, in cooperation with UFSCar.
Figure 1: crystalline metals structure versus metallic glasses structure
Surface fontionalization and corrosion resistance of coating
In relation with metallic glass studies, we also work on corrosion resistance of amorphous and nanocrystalized (partially crystalized) coatings for biomedical applications. For instance, we deposed Fe or Ti based amorphous or nanocrystallized metals on steel substrates.
The recent acquisition of a “Local Electrochemical Impedance Spectroscopy (LEIS)” device enables local characterization to highlight heterogeneous reactivity along the surface (defects, porosity …). The measurements consist in collecting a local current through a bi-electrode which moves along the surface (3D motion) thanks to piezoelectric motors. Figure 2 shows the local electrochemical impedance measurement system. Coatings of Ti and TiN have been studied for biomedical applications. Thanks to PVD technique, stainless steels have been covered by TiN or Ti layers (in collaboration with SIMaP laboratory and Ugitech company). Coatings corrosion properties, and more specifically pitting corrosion, are studied in physiological media and compared to the ones of stainless steel substrate.


Figure 2: local electrochemical impedance measurement system (the complete device on the left and the bi-electrode on the right)
Surface functionalizartion and modeling of complex interfaces by EIS
We also focused on the electrochemical behavior of surfaces and interfaces in the media with which they interact. In this framework, anodized TiO2 nanotubes (see figure 3) layers with a very specific topology were studied as “model surfaces”. We characterize them by Electrochemical Impedance Spectroscopy (EIS) in physiological media in order to cross topological information and electrochemical characterizations.
Thanks to an alternative representation of impedance diagrams proposed by our team, we showed that the electrochemical behavior of this material is complex and should be modeled by a three-time constant equivalent circuit rather than the two-time constant equivalent circuit usually proposed in the literature.
In addition to that, the combination of good mechanical properties, excellent corrosion resistance, high specific surface area and biocompatibility exhibited by this nanostructured material enables its extensive use in biomedical devices, for implants for example (and more specifically for hip replacement or tooth screws). This study is carried out in collaboration with a Brazilian laboratory, NanoBio, from the university of Sao José dos Campos (Univap).

Figure 3: Top and cut view of TiO2 nanotubes synthesized by anodization
Corrosion monitoring -Electrochemical noise (Pr. Ricardo Nogueira)
In LEPMI we focus on the “Electrochemical Noise” technique. Its principle is to measure the spontaneous fluctuation of the current (or potential) passing through an interface under potentiostatic (or galvanostatic) control. The analysis of electrochemical noise can give useful information about the rate and nature of the chemical processes taking place at the electrodes, corrosion for example.